|
Download View publication Copy reference Copy caption Embed figure Ternary phase diagram as predicted by FactSage 7.0 software for CaO-Al 2 O 3SiO 2-TiO 2, detailing liquid slag phase at 1350C and 1400C as well as compositions for starting bauxite residue and final slag.Source publication Combined SAF Smelting and Hydrometallurgical Treatment of Bauxite Residue for enhanced Valuable Metal Recovery Conference Paper Full-text available Oct 2017 Bengi Yagmurlu Gzde Alkan Buhle Sinaye Xakalashe.Carsten Dittrich Reductive smelting trials were undertaken on a bauxite residue (red mud) sample via SAF.
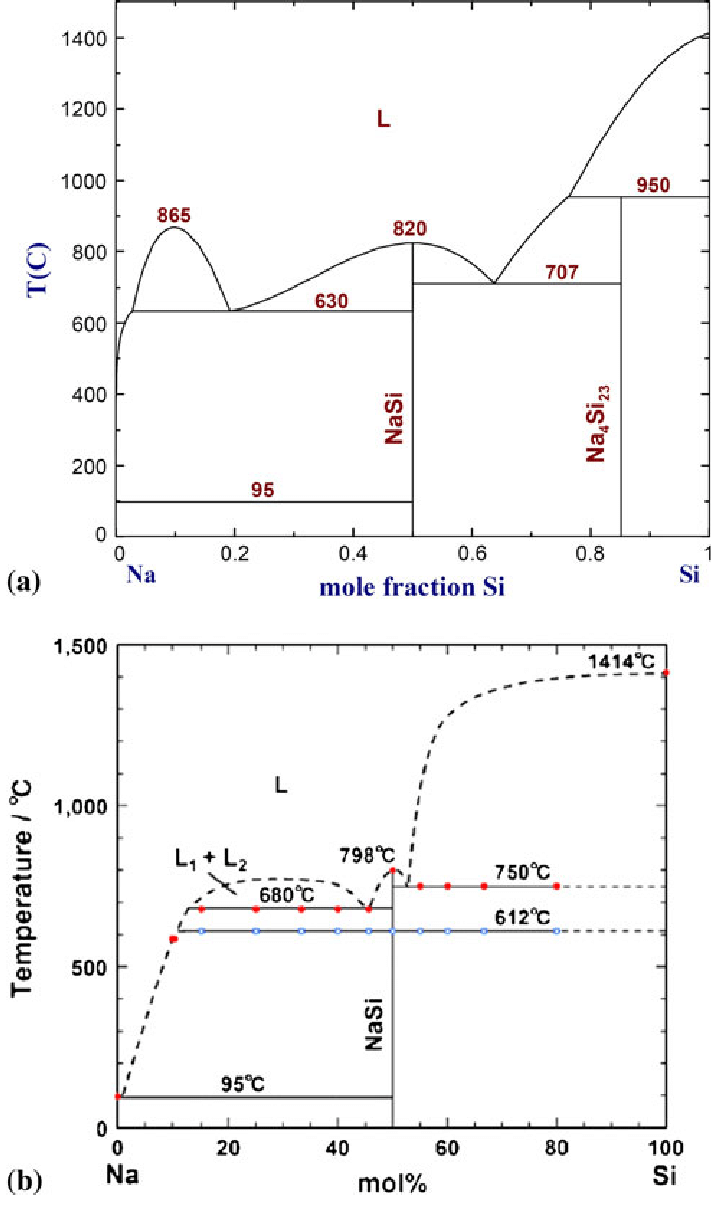
Factsage Software Software For CaOLignite coke was the selected reductant and CaO was used for fluxing achieving low operating temperatures (1500 1550 C) and good slag-metal. Cite Download full-text Context in source publication Context 1. XRD patterns below show clearly that the bauxite residue undergoes a transformation during the smelting process. The gehlenite phase (2CaO.Al 2 O 3.SiO 2 ) was formed during the smelting and cooling process as confirmed by both the slag XRD results in Figure 3 and the phase diagram in Figure 2. Titanium is contained in the perovskite (CaTiO 3 ) phase which would have crystallised during the relatively slow cooling of the slag, the formation of this perovskite phase is expected to have an effect on the subsequent recovery of Ti via leaching.. View in full-text Similar publications Zinc recovery from metallurgical slag and dust by coordination leaching in NH 3 CH 3 COONH 4 H 2 O system Article Full-text available Jul 2018 Aiyuan Ma Xuemei Zheng Song Li. 
This paper describes the production feasibility and rationality of a cleaner zinc recovery process using MSD and a hydrometallurgical method. View 7 Recovery of rare earth elements from phosphogypsum Article Full-text available Jun 2018 V.N. Rychkov Evgeny Kirillov Sergey Kirillov. A.s. Malyshev Phosphogypsum is a common large-tonnage waste of phosphoric fertilizers industry. Joint co-crystallization ofREE with gypsum provided a low degree of REE leac. View Leaching of lead and copper from flash smelting slag by citric acid Article Full-text available May 2019 Krzysztof Gargul Boena Boryczko Andelika Bukowska. Stanisaw Maecki As a direct-to-blister copper flash smelting slag contains important levels of copper (14) and lead (24) it is subjected to a process of high temperature reduction. To recover copper as well as lead the slag is subjected then to a process of decopperization by reducing the oxide metal compounds in an electric furnace. Six different slags generated by reductive smelting of the same bauxite residue sample were treated by high-pressure acid leaching (HPAL) with HCl and H2SO4 to selectively extract REEs. The extraction of yttrium, lanthanum and neodymium was above 95 wt when HCl was used as a reagent, but it was much lower ( 90 wt, 18 g L1), while the concentration of the remaining iron (60 wt) was of 3 g L1 in the leachate. The co-dissolution of silicon and titanium was lower than 5 wt. Download lagu gendang silat mp3View. Fe(III) was selectively removed by a dual-step precipitation procedure (Yagmurlu et al., 2017a (Yagmurlu et al.,, 2017b. First, the largest part of Fe (III) was precipitated by addition of 10 wt NH 3(aq), until the pH of the solution reached the value of 3.3 and then the mixture was filtered... Selective removal of Fe(III) was attempted in order to increase the efficiency of the SILP column chromatography operation for REEs (and especially Sc) purification from H 2 SO 4 medium. The SILP betainium sulfonyl(trifluoromethanesulfonylimide) poly(styrene-co-divinylbenzene) Hbet-STFSI-PS-DVB exhibits a high selectivity for the rare-earth elements (REEs) over other elements in acidic BR leachates. However, scandium(III) uptake by the Hbet-STFSI-PS-DVB from sulfuric acid leachates is difficult due to strong electrostatic interactions of small scandium(III) ions with sulfate anions. Sulfuric acid leachates generally contain high concentrations of base metal ions like iron(III) and this reduces the efficiency of the Hbet-STFSI-PS-DVB for uptake of REEs. Asus tf101 upgrade to 70Therefore, a precipitation step for iron(III) removal with aqueous ammonia solution was introduced, as a simple and economically viable pretreatment step of BR leachate, prior to the REEs recovery by the SILP. Iron(III) precipitation from sulfuric acid BR leachates increased the efficiency of purification by column chromatography.
0 Comments
Leave a Reply. |
AuthorSusan ArchivesCategories |
 RSS Feed
RSS Feed
